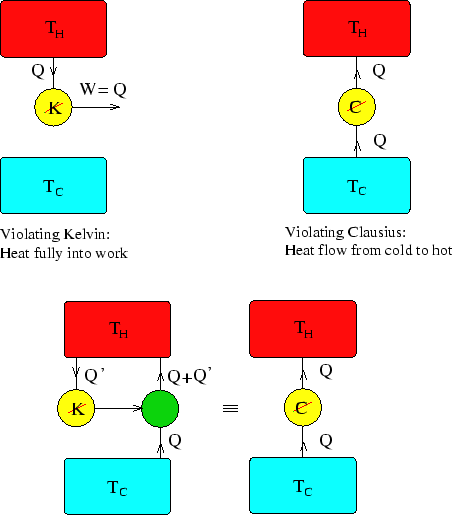
The latter is only laid down as the fundamental principle for deriving the theorem of the equivalence of transformations. Clausius’ mechanical theory of heat, published in the nineteenth century, is then introduced and discussed in detail, from which it is found that Clausius himself regarded “Theorem of the equivalence of the transformation of heat to work, and the transformation of heat at a higher temperature to a lower temperature”, rather than “Heat can never pass from a colder to a warmer body without some other change”, as the statement of the second law of thermodynamics. There are some phenomena in other disciplines similar to the Clausius Statement of the second law, but none of them has been accepted as the statement of a certain law. 
In this paper, we first analyze the difference between the second law of thermodynamics and the laws in other disciplines.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
